從LoMei入行以來,總會聽到這個莫名其妙的10%說法,為什麼說是莫名其妙呢?因為在「無菌製劑」的製程放大執行參數,以混和階段為例:10L和50L(x5)的混和強度/時間就存在差異,何況是100L(x10)?但這都只是「說法」爾已,通常對於無菌製劑批量改變這件事都還是會重新做製程確校去確認各製程參數是否符合批量。
那這個10%理論到底從何而來?以下列出跟10%、x10相關文件。(先說結論:批次相關的10%、x10通常發生在口服製劑-LoMei沒有實務經驗,和無菌製劑的相關性極低;但無論是什麼製劑種類,查驗登記後的更改批量都是需要經過嚴謹的評估。)
美國US
- SUPAC-MR: Modified Release Solid Oral Dosage Forms
Scale-Up and Postapproval Changes: Chemistry, Manufacturing, and Controls; In Vitro Dissolution Testing and In Vivo Bioequivalence Documentation
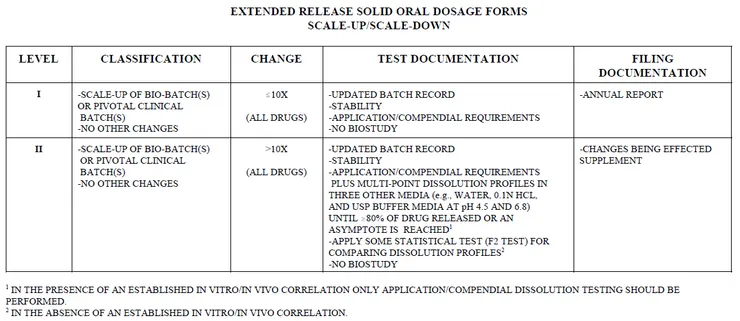
Scale-Up and Postapproval Changes: Chemistry, Manufacturing, and Controls; In Vitro Dissolution Testing and In Vivo Bioequivalence Documentation
歐洲EU
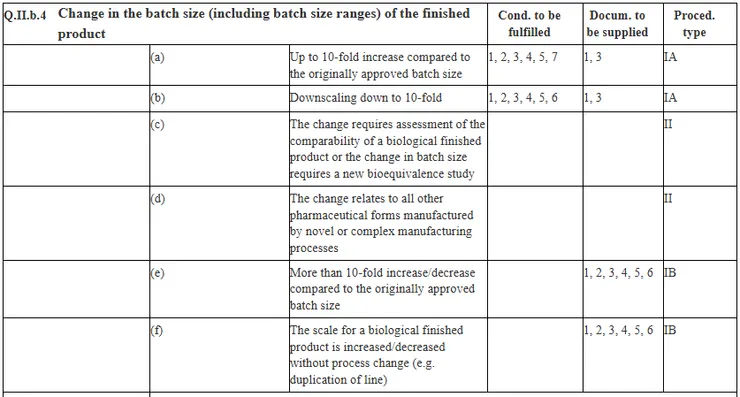
- 歐洲藥品管理局 EMA - Guideline on process validation for finished products - information and data to be provided in regulatory submissions
- Communication from the Commission – Guidelines on the details of the various categories of variation, on the operation of the procedures laid down in Chapters II, IIa, III and IV of Commission Regulation (EC) No 1234/2008 concerning the examination of variations to the terms of marketing authorisations for medicinal products for human use, and on the documentation to be submitted pursuant to those procedures
東協 ASEAN
- ASEAN Variation Guideline for Pharmaceutical Products Revision 2
以下的截圖是某次遇到差不多的問題時,LoMei問小主管得到的回答。